 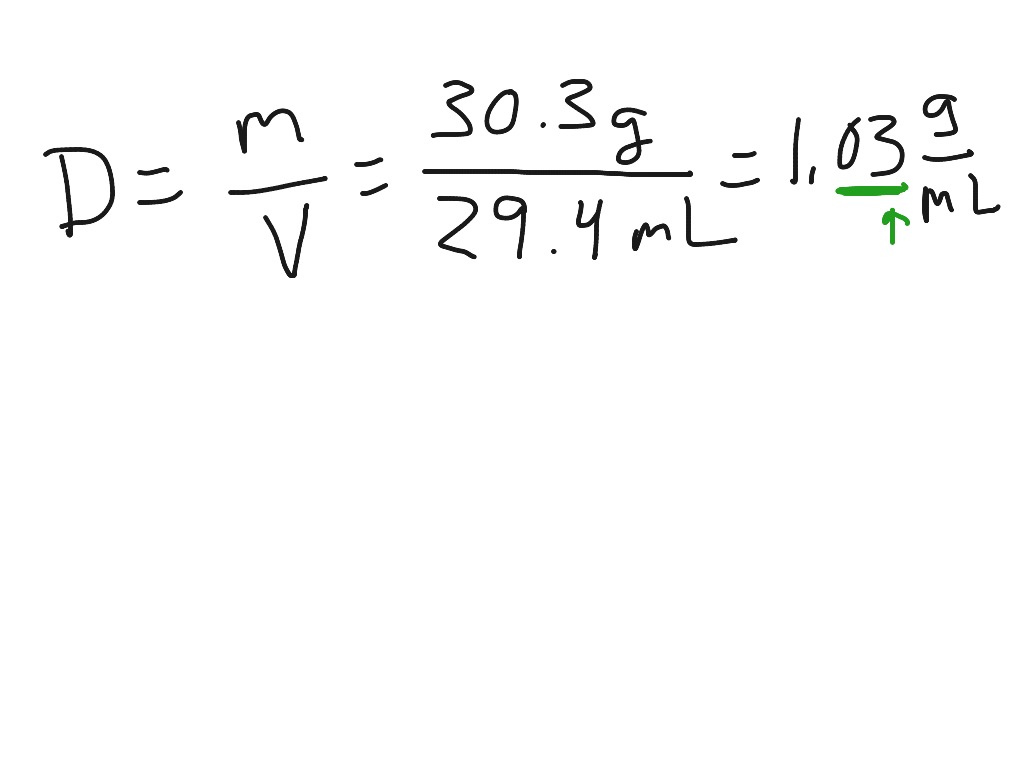
Water is most dense at 4 ☌ and at that temperature has a density of 1 g/cm3. There are exceptions however, such as water's density increasing between 0☌ and 4☌.īelow is a table of units in which density is commonly expressed, as well as the densities of some common materials. density of water changes with temperature. Increases in temperature tend to decrease density since the volume will generally increase. 
Aber this is not the end Performing your own experiment at home use a less objects with somebody unknown seal (but more or less equal to this value for the density of water, i.e., 1000 kg/m³ ). The same density of water is 1.0249 g/ml conversely 63.982 lb/ft³. An increase in pressure decreases volume, and always increases density. The density of salt water is 1,024.9 kg/m³. However, when regarding gases, density is largely affected by temperature and pressure. In this paper, a new formulation of the density of water (based primarily on the work of Kell 2) as a function of temperature on the 1990 International Temperature Scale is presented. Below is a table of units in which density is commonly expressed, as well as the densities of some common materials. In many quarters, the formulation of Wagenbreth and Blanke 1 is used to calculate the density of water. There are exceptions however, such as water's density increasing between 0C and 4C. In the case of solids and liquids, the change in density is typically low. Increases in temperature tend to decrease density since the volume will generally increase. Note that density is also affected by pressure and temperature. It is useful to carefully write out whatever values are being worked with, including units, and perform dimensional analysis to ensure that the final result has units of mass There are many different ways to express density, and not using or converting into the proper units will result in an incorrect value. However, it is important to pay special attention to the units used for density calculations. The calculation of density is quite straightforward. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
